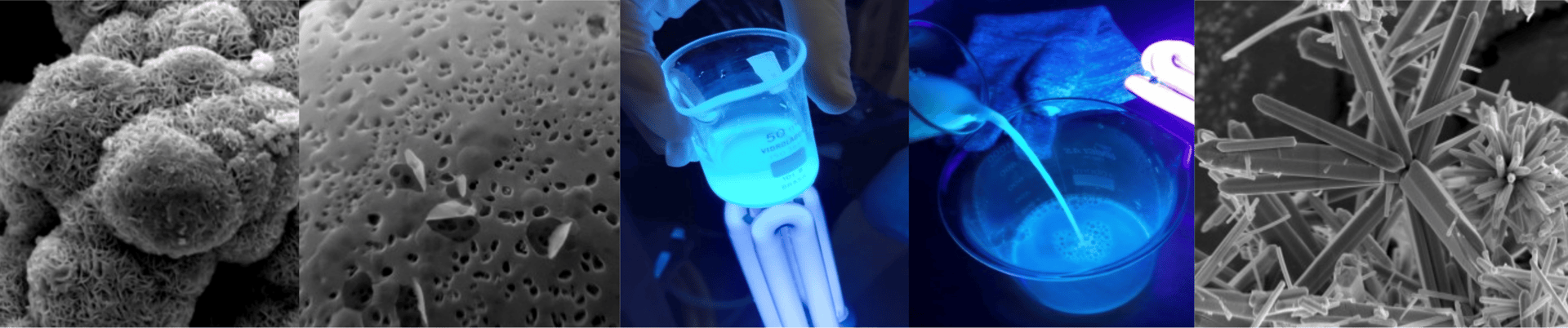
Activated carbon (AC) is a porous material generally produced from carbon-rich precursors that undergo an activation process to develop a very large internal pore area. This structure functions as a kind of “molecular sponge”, allowing activated carbon to adsorb a wide variety of substances. For this reason, it is widely used in water and air filtration, wastewater treatment, industrial processes, and even pharmaceutical applications.
At GMCM, we study high-surface-area activated carbons for the adsorption of contaminants at solid–gas and solid–liquid interfaces, as well as their use as supports for semiconductors. More recently, the group has also been applying activated carbons in energy storage systems (capacitance-based devices).


Activated Carbon


Carbon quantum dots (CQDs) are carbon-based nanomaterials (typically < 10 nm) that exhibit fluorescence and high stability. Due to their small size and surface chemistry, they can interact with different chemical species and are widely applied in sensors, bioimaging, photocatalysis, and functional materials.
At GMCM, we study CQDs combined with nanostructured semiconductors (such as high-entropy perovskites, zinc oxide, niobium oxide, etc.) to investigate exciton lifetimes in photocatalytic processes.
Carbon Quantum Dots
Nanostructured Semiconductors


Semiconductor nanomaterials are nanoscale structures that exhibit tunable electronic and surface properties, enabling them to act as active materials in catalytic processes. Due to their small size and high specific surface area, these materials promote interactions with different chemical species and are widely applied in heterogeneous catalysis and functional materials.
At GMCM, we study semiconductor nanomaterials such as high-entropy perovskites and oxides of iron, zinc, niobium, and tungsten, focusing on catalytic applications. To optimize dispersion and maximize the active surface area, we use activated carbons as supports, taking advantage of their high porosity and surface chemistry to enhance the interaction between catalytic phases and the reaction medium. This strategy enables the development of more efficient, stable, and functionally versatile materials, expanding their potential in catalytic processes and advanced materials.