Optimization of chemically activated carbon derived from malt bagasse for CO2 adsorption: a simplex-centroid approach. Environmental Science and Pollution Research
2026
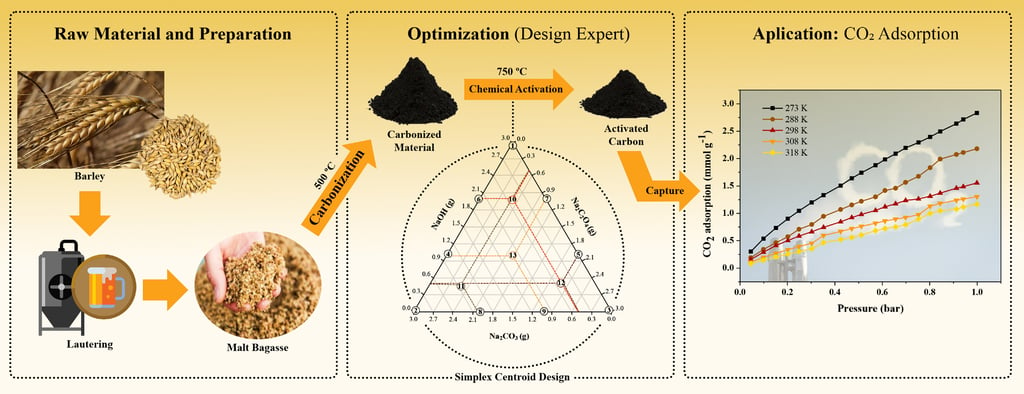
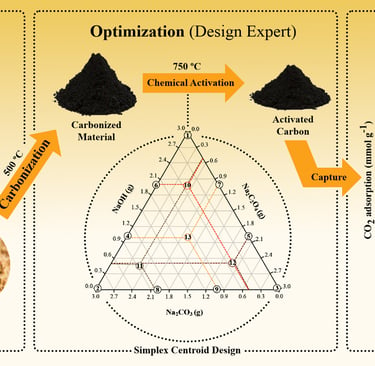
The intensification of the greenhouse effect has been primarily driven by anthropogenic CO₂ emissions, leading to significant climate change. Among the strategies to mitigate CO₂ emissions from industrial activities, adsorption using activated carbons (ACs) derived from renewable sources stands out as a promising approach. Activated carbons were prepared via chemical activation of malt bagasse, a brewing industry by-product, using NaOH, Na₂C₂O₄, Na₂CO₃, and their mixtures as activating agents. The augmented simplex-centroid method was employed to optimize the process and obtain an activated carbon (ACₒₚ) with high BET surface area. The materials were characterized by proximate analysis, N₂ physisorption, thermogravimetric analysis (TGA), scanning electron microscopy (SEM), Fourier-transform infrared spectroscopy (FTIR), and Raman spectroscopy. CO₂ adsorption experiments were performed at different temperatures to assess performance. The optimized carbon (ACₒₚ) exhibited a well-developed porous structure and high surface area, achieving a CO₂ adsorption capacity of 2.83 mmol g⁻1 at 273 K. The results demonstrated that NaOH-activated carbon provides favorable properties for CO₂ capture, confirming the feasibility of malt bagasse as a sustainable precursor for efficient adsorbent materials.